Medgadget: The pandemic has changed how many people visit with their doctors, with more remote interactions and virtual visits. Early response from customers has been overwhelmingly positive and we look forward to growing our market share in this segment over the coming years. Vanta is a high-performance recharge-free neurostimulator that can be paired with our latest therapy, DTM SCS endurance therapy, to offer patients meaningful pain relief and an estimated 5.5-7.5 years of longevity based on actual programmed settings from our study. How are you looking at this space and patient population?Ĭharlie Covert: While it’s true that we have not historically made a lot of noise in the recharge-free segment of this market, which represents about a quarter of all chronic pain patients, that is no longer the case with the launch of our FDA-approved Vanta™ neurostimulator this past summer. Medgadget: Patients have an option between rechargeable and recharge-free implantable spinal cord stimulators, but historically Medtronic has not been a major player in the recharge-free segment of this market. Data has shown DTM™ SCS therapy with the Intellis™ platform provides superior, sustained, and profound back pain relief. Unlike conventional SCS, Differential Target Multiplexed (DTM) stimulation targets different areas of the spinal cord – hence, Differential Target – with different signals or frequencies-hence, Multiplexed. Our randomized, controlled trial for DTM SCS, the results of which were published in Pain Practice Journal, showed 84% of chronic back pain patients treated with DTM™ SCS reported at least 50% pain relief, compared with only 51% of conventional SCS patients at 12 months. Perhaps our most consequential new technology in this space is DTM™ SCS therapy, which coordinates multiple signals at multiple anatomical targets and is proven superior to conventional SCS. Features like more efficient, longer lasting batteries, smaller and more contoured devices for comfort, broader MRI access, more robust data reporting for physicians, and personalized stimulation thanks to AdaptiveStim™ technology, which automatically adjusts stimulation based on the patient’s body positions. We have also made strides in recent years to take this technology further. Medtronic pioneered spinal cord stimulation more than a 40-years ago. How has the technology evolved in recent years? What’s new?Ĭharlie Covert: Chronic pain is a debilitating condition, and symptoms can persist over years for many patients. Medgadget: Spinal cord stimulation is a chronic pain therapy that has been around for several decades. Implantable Pain Therapies is part of our Neuromodulation operating unit.  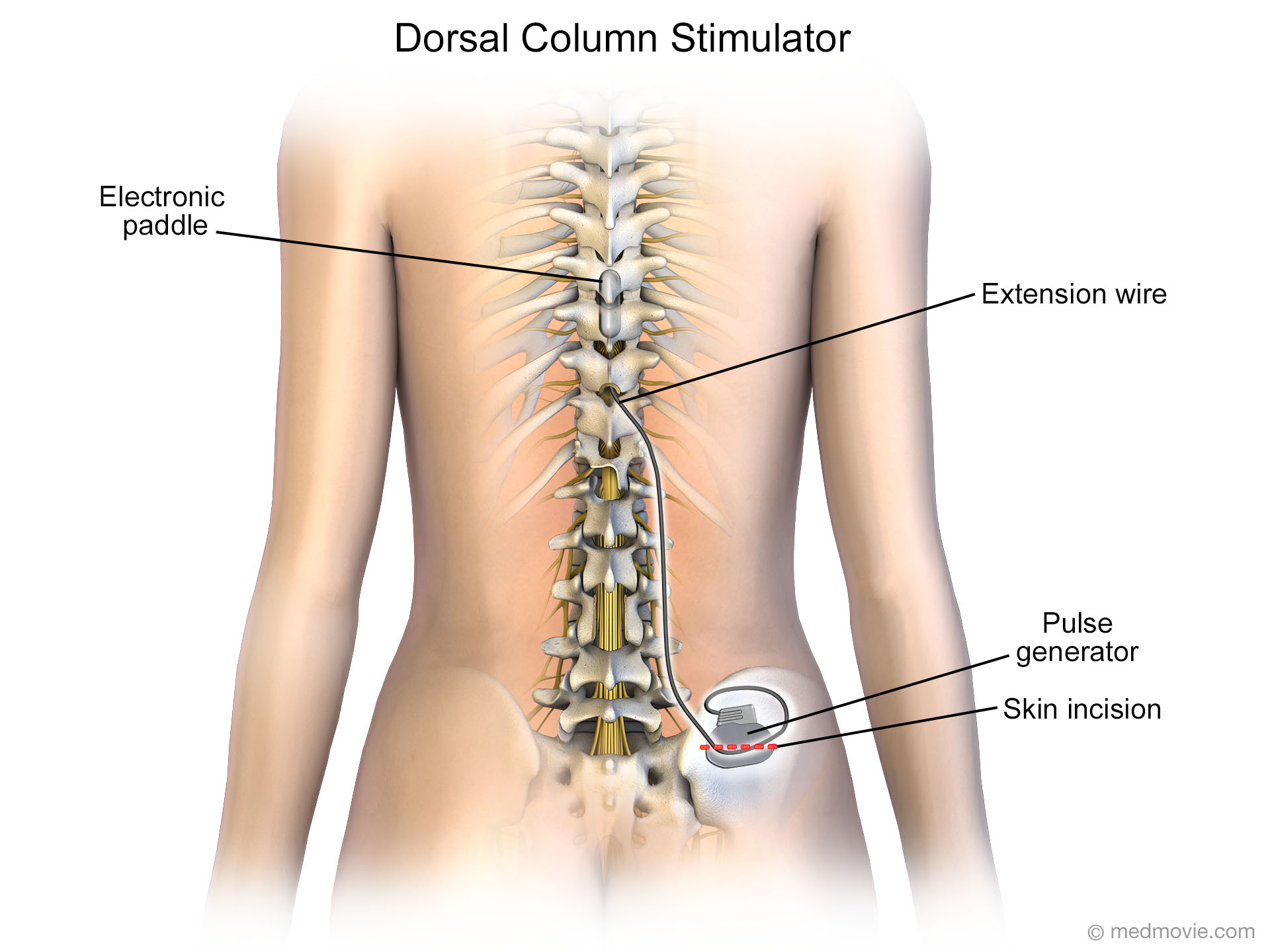
In 2018, I assumed the role of General Manager for our Implantable Pain Therapies business, which is comprised of our spinal cord stimulation (SCS) and targeted drug delivery (TDD) technologies. I also led product planning for our first Bluetooth-enabled platforms in that business. Prior to my current role, I led Medtronic’s targeted drug delivery business, and prior to that, led marketing and strategy for our pacemaker business that launched the Micra transcatheter pacemaker. I started working for Medtronic over 15 years ago, spending the majority of my time in commercial, strategy, and business development roles. Charlie Covert, Medtronic: My experience in the medical device industry spans about two decades.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
